SUMMARY
This is AI generated summarization, which may have errors. For context, always refer to the full article.
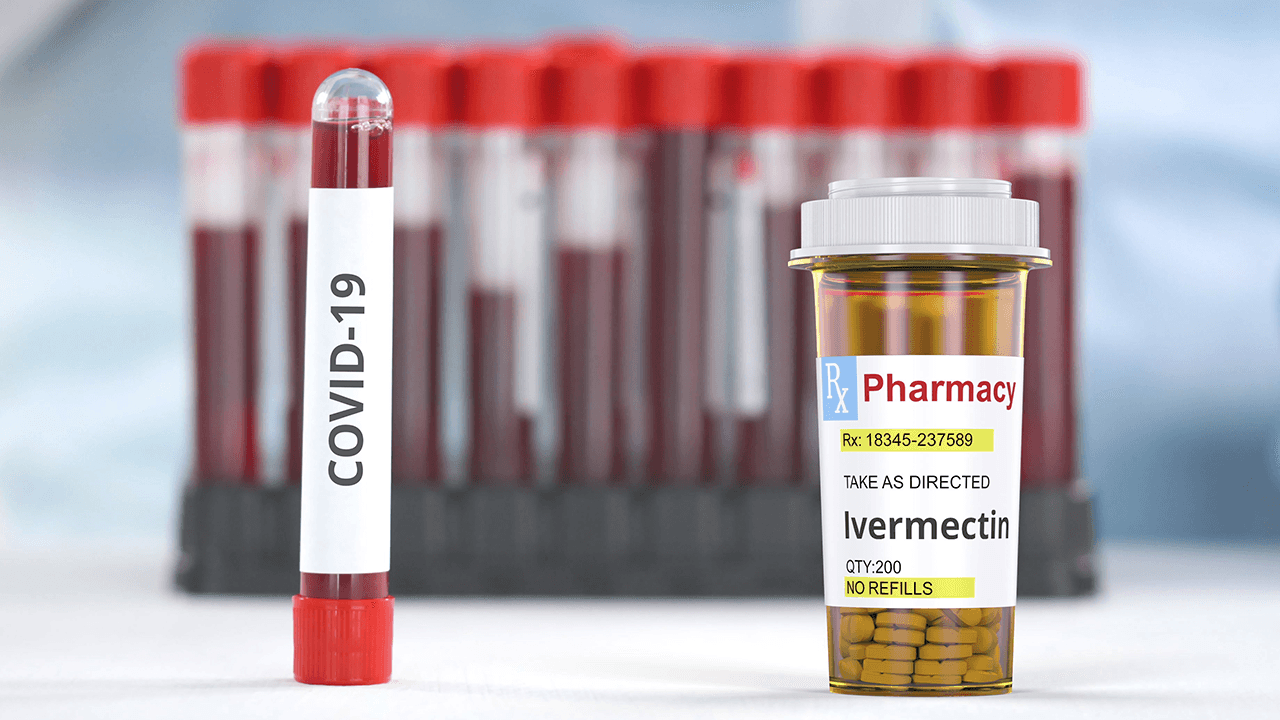

The Department of Health (DOH) said on Monday, April 5, that the Philippines’ Food and Drug Administration (FDA) is “currently processing an application for product registration” of anti-parasitic drug Ivermectin for human use in the country.
In a statement sent to media, the DOH said “concerned parties” filed the application on March 31, without identifying who they were.
“All current Ivermectin products registered in the country are for veterinary use and are only allowed for the treatment of internal and external parasites as well as prevention of heartworm disease in animals,” the DOH said.
This latest statement from the DOH comes amid growing discussions over the use of Ivermectin to treat COVID-19.
The issue was brought to light after doctors in the Philippines and abroad, as well as a number of lawmakers, urged the DOH and the FDA to hasten the process of approving Ivermectin as a COVID-19 treatment in the country.
In a House health panel on March 30, doctor Allan Landrito – the most popular proponent for Ivermectin as COVID-19 treatment in the Philippines – said he made Ivermectin pills on his own before selling thousands of bottles across the country.
In the same hearing, World Health Organization (WHO) Representative to the Philippines Dr Rabindra Abeyasinghe stressed the need for “carefully controlled and planned” clinical trials of Ivermectin before it could be used to treat COVID-19 in the country.
DOH still doesn’t recommend Ivermectin
While the DOH said that it had received the application for registration of Ivermectin, it maintained its position that it “does not recommend the use” of the anti-parasitic drug to treat COVID-19.
The DOH again cited the joint statement of 10 medical societies in the Philippines, including the Philippine Society of Public Health Physicians and the Philippine Society of Microbiology and Infectious Diseases, saying that they do not recommend the use of Ivermectin for COVID-19 treatment.
In their statement, the groups cited a systematic review of controlled trials that showed Ivermectin did not significantly reduce the risk of mortality among COVID-19 patients.
“Based on the current evidence from randomized controlled trials, we do not recommend the use of Ivermectin for the treatment of COVID-19,” they said.
Meanwhile, the DOH clarified that while the FDA approved Chinese traditional medicine Lianhua Qingwen, it can only be used with a doctor’s prescription. It added that the drug is not registered as a COVID-19 medication.
To date, there is still no approved treatment for COVID-19. What hospitals are using now are investigational drugs allowed under the compassionate use permit of the FDA. – Rappler.com
Add a comment
How does this make you feel?







![[PANOORIN] Naku! Mag-ingat sa unregistered health products online!](https://www.rappler.com/tachyon/2023/05/fact-check-ls-3.jpg?resize=257%2C257&crop=283px%2C0px%2C720px%2C720px)





![[OPINION] On Ivermectin, COVID-19 response, and the business sector](https://www.rappler.com/tachyon/2021/05/tl-sq-1.jpg?resize=257%2C257&crop_strategy=attention)
There are no comments yet. Add your comment to start the conversation.