SUMMARY
This is AI generated summarization, which may have errors. For context, always refer to the full article.
The Philippine Food and Drug Administration (FDA) has approved the Department of Health’s application for an emergency use authorization (EUA) covering Sinopharm COVID-19 vaccines donated to the government.
This was announced by FDA Director-General Eric Domingo on Monday, June 7, during a meeting with President Rodrigo Duterte in Davao City.
“Our experts also looked at it (Sinopharm vaccines) and our evaluation from FDA, and today we already granted an emergency use authorization to DOH to accept the donations of Sinopharm,” said Domingo.
On May 20, the Department of Health (DOH) submitted an EUA application for the Sinopharm vaccine. The EUA process allows for a government agency to apply for this crucial label. Sinopharm representatives failed to provide all the requirements needed for the FDA to complete its review of their vaccine.
The DOH move also came after the World Health Organization approved for emergency use Sinopharm’s jab, a sign in the vaccine’s favor that health officials had said would likely speed up its EUA process in the Philippines.
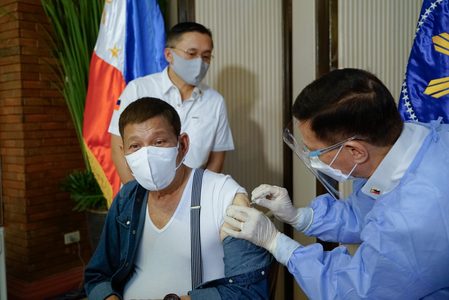
In early May, Duterte was administered a first dose of the Sinopharm vaccine even if it was not covered by any Philippine EUA, prompting critics to say the President was cutting corners while his administration told regular Filipinos they couldn’t choose their vaccine.
Duterte’s vaccination was covered only by a compassionate special permit issued for 10,000 Sinopharm doses intended for his security guards, the Presidential Security Group.
Malacañang later on said that a separate batch of 1,000 Sinopharm doses was donated to the Philippines and that one of those doses was the one Duterte received.
The government is expecting to enter into more talks with Sinopharm and the Chinese government to obtain more doses. – Rappler.com
Add a comment
How does this make you feel?


![[Time Trowel] Evolution and the sneakiness of COVID](https://www.rappler.com/tachyon/2024/02/tl-evolution-covid.jpg?resize=257%2C257&crop=455px%2C0px%2C1080px%2C1080px)


There are no comments yet. Add your comment to start the conversation.