SUMMARY
This is AI generated summarization, which may have errors. For context, always refer to the full article.
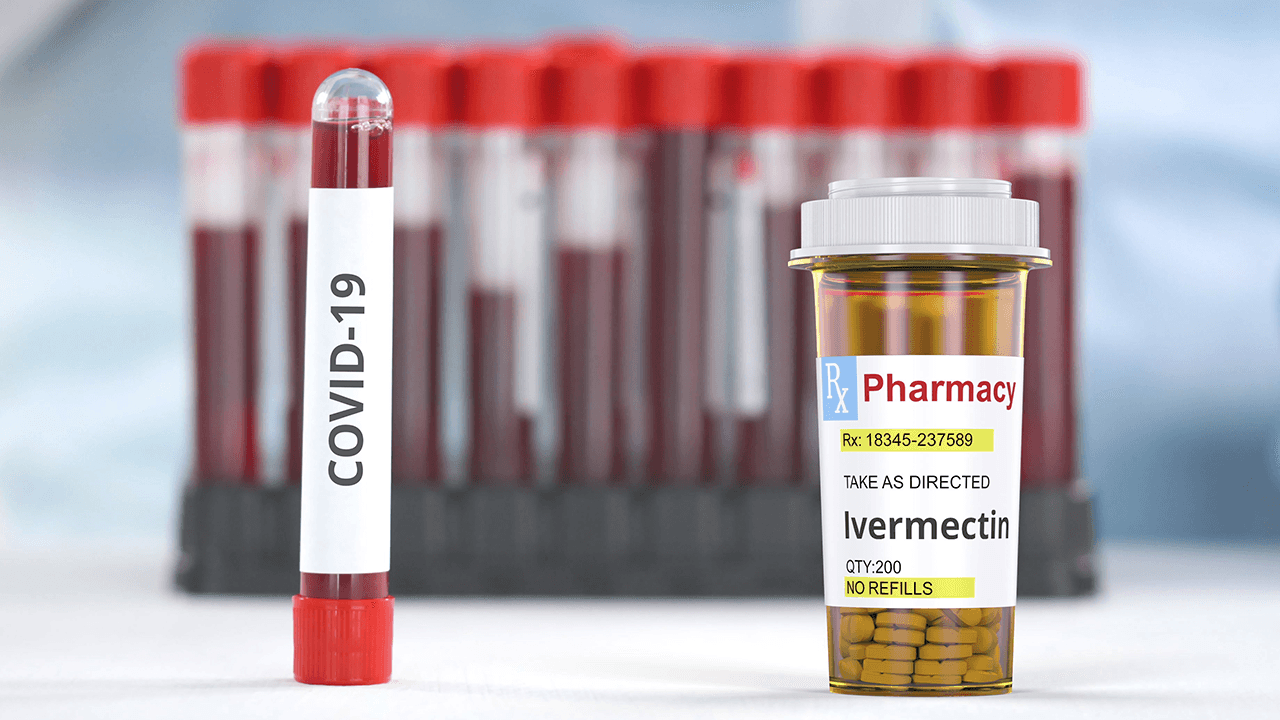
The Philippines’ Food and Drug Administration (FDA) on Thursday, April 8, issued a limited permit for an undisclosed hospital to use the anti-parasitic drug Ivermectin as treatment for COVID-19.
“Mayroon na po kaming na-grant dahil ang Ivermectin ay isang investigational product, at alam natin may mga clinical trials ongoing dito para gamitin against COVID-19,” FDA Director General Eric Domingo said in the Laging Handa press briefing aired on state-run PTV-4.
(We have granted [a compassionate use permit] because Ivermectin is an investigational product, and we know that there are ongoing clinical trials on using this drug against COVID-19.)
“May isang ospital sa amin na nag-apply for compassionate special use permit at na-grant na nga ng araw na ito,” he added. (There is one hospital which applied for compassionate use permit, and we granted it today.)
In a text message to Rappler, Domingo said he could not give the name of the hospital “out of patients’ privacy.”
A compassionate special permit (CSP) allows for the limited use of investigational drugs or unregistered drugs.
According to a 1992 FDA administrative order, a hospital or doctor can seek access to an investigational drug through a CSP for patients with life-threatening conditions when there is “no existing superior alternative therapy that can likely cure or adequately control their conditions.”
The use of the drug is also limited to the hospital to which the permit is granted, which means it cannot be sold in the market.
Having this permit does not guarantee either that the drug is proven effective or already recommended by the FDA.
To date, there is still no approved treatment for COVID-19. Hospitals are using investigational drugs allowed under the compassionate use permit – Ivermectin now one of them in this unnamed hospital.
There is an ongoing public debate over the use of Ivermectin to treat COVID-19 – with some doctors recommending it and patients sharing how it had been beneficial to them, while other medical experts warned against its unproven use against the coronavirus diseases.
In the Philippines, the Ivermectin allowed – and freely sold online – is the one for animals, used to treat heartworm disease. For humans, the Ivermectin available in the country is “in topical formulations under prescription use only,” the FDA had said.
Need for registration, process
Domingo said that there are two local manufacturers in the country applying for certificate of product registration of Ivermectin, stressing the need for the product to undergo registration process to assure quality and safety.
In a House committee on health hearing on March 30, Dr Allan Landrito – the most popular proponent of Ivermectin for COVID-19 treatment in the Philippines – said he had made Ivermectin pills for at least 8,000 patients who were begging him to treat them. He stopped selling Ivermectin when the FDA warned about its sale without permits.
In the same hearing, World Health Organization Representative to the Philippines Dr Rabindra Abeyasinghe stressed the need for “carefully controlled and planned” clinical trials of Ivermectin before it could be used to treat COVID-19 in the country. – with reports from Sofia Tomacruz/Rappler.com
Add a comment
How does this make you feel?


![[Time Trowel] Evolution and the sneakiness of COVID](https://www.rappler.com/tachyon/2024/02/tl-evolution-covid.jpg?resize=257%2C257&crop=455px%2C0px%2C1080px%2C1080px)





![[PANOORIN] Naku! Mag-ingat sa unregistered health products online!](https://www.rappler.com/tachyon/2023/05/fact-check-ls-3.jpg?resize=257%2C257&crop=283px%2C0px%2C720px%2C720px)

There are no comments yet. Add your comment to start the conversation.