SUMMARY
This is AI generated summarization, which may have errors. For context, always refer to the full article.
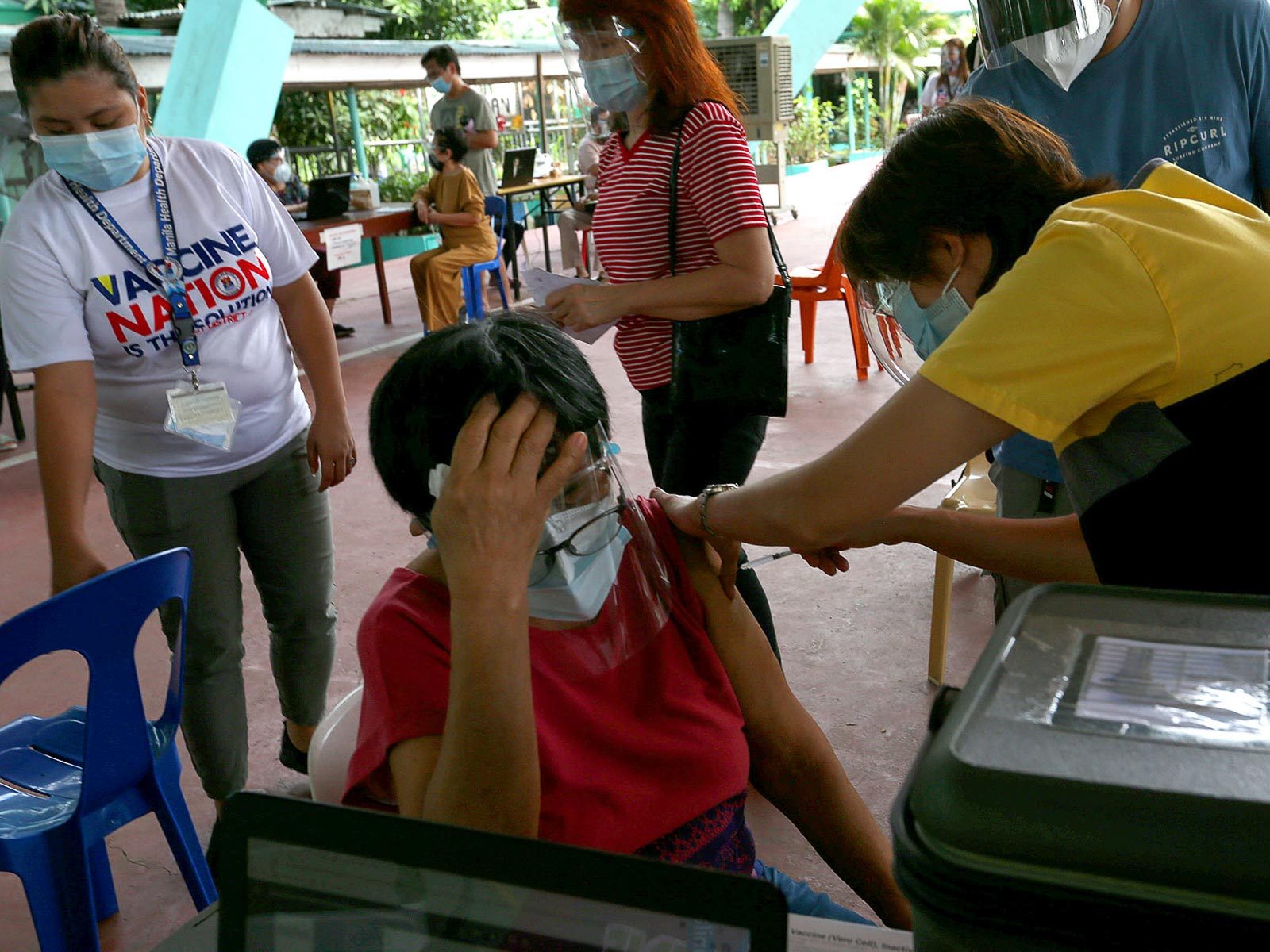
More local government units (LGUs) in Metro Manila began on Monday, April 12, to use the COVID-19 vaccine from Chinese drug manufacturer Sinovac to inoculate their elderly population.
When Sinovac was first granted an emergency use authorization (EUA) for its vaccine in February, senior citizens were excluded from the list of people who were allowed to receive its vaccine, due to its varying efficacy rate in trials in Brazil and Indonesia.
But the the Food and Drug Administration (FDA) and the Department of Health (DOH) on April 7 gave the green light to LGUs to administer CoronaVac to senior citizens amid the shortage in AstraZeneca vaccine supplies.
Government health experts also recommended to expand Sinovac’s use after data from the first two phases of trials showed the vaccine could safely trigger an immune response for senior citizens.
On Monday, San Juan kicked off the rollout of the CoronaVac for their elderly population, saying their supply of AstraZeneca vaccine doses from global vaccine sharing alliance COVAX facility had run out.
“Hindi tayo puwedeng tumigil sa pagbabakuna ng senior citizens dahil lang naubusan tayo ng AstraZeneca (We cannot stop the vaccination of senior citizens just because our Astrazeneca vaccine supply ran out),” he said.
Zamora added that April 12 “is really the start of the vaccination of senior citizens using Sinovac” based on Advisory No. 27 of the National COVID-19 Vaccination Operations Center.
The Manila and Quezon City LGUs already began administering CoronaVac for senior citizens last week, after the Department of Health on April 7 issued Memorandum 2021-0175, which detailed additional guidelines for administering Sinovac’s vaccine.
Senior citizens in Muntinlupa and Parañaque were also scheduled on Monday to receive a vaccine dose from Sinovac.
While the Philippines has yet to replenish its supply of Astrazeneca vaccines, the government received on Sunday 500,000 more doses of CoronaVac.
FDA Director General Eric Domingo last week listed numerous reasons why it approved the use of Sinovac’s vaccine on the elderly.
Among them is the fact that the Sinovac jab is currently the most accessible option available for Filipinos amid the unprecedented spike in COVID-19 cases in the Philippines.
The FDA however asserted that a “stringent evaluation” of a person’s health and risks should be made to determine whether they could be administered with CoronaVac. – Rappler.com
Add a comment
How does this make you feel?




![[OPINYON] Puting buhok at ang mapaminsalang ageism](https://www.rappler.com/tachyon/2024/03/IMHO-Ageism-March-11-2024.jpg?resize=257%2C257&crop=468px%2C0px%2C720px%2C720px)




There are no comments yet. Add your comment to start the conversation.