SUMMARY
This is AI generated summarization, which may have errors. For context, always refer to the full article.

After weeks of delay, the Philippines finally started its COVID-19 vaccination drive, among the last to do so in Southeast Asia.
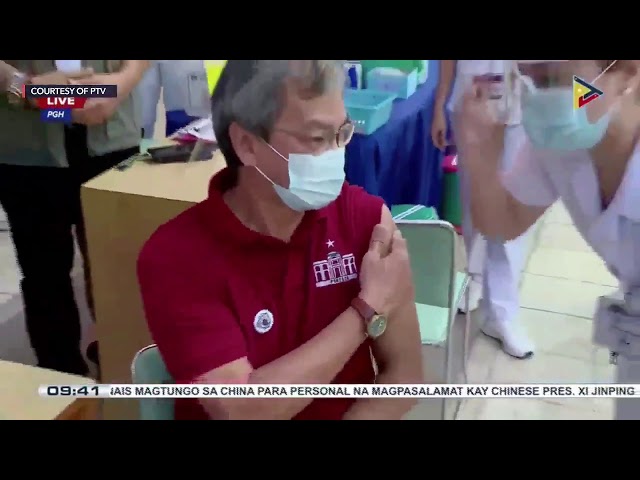
Philippine General Hospital (PGH) Director Gerardo “Dr Gap” Legaspi was the first to roll up his sleeves as he received his shot of CoronaVac developed by Chinese company Sinovac.
Legaspi is also the first health worker in the country who received a legal COVID-19 vaccine following the unauthorized vaccination of several high-ranking government officials and close-in security personnel of President Rodrigo Duterte.
PGH nurse Chareluck Santos administered the vaccine shot to Legaspi.
Here’s what we know about Legaspi:
- Legaspi has been into active practice and teaching in neurosurgery for 25 years now.
- In the Philippines, he pioneered awake craniotomy surgery, a brain surgery performed on patients who are awake.
- His expertise includes treating aneurysms, arteriovenous malformation, brain tumors, congenital malformations of the brain and spinal cord, and skull base surgery.
- He is the former president of the the ASEAN Neurosurgical Society and the Academy of Filipino Neurosurgeons Inc.
- He has clinics in PGH, St Luke’s Medical Center, and the Medical City.
- He also works as a consultant in other government institutions as well, like Philippine Children’s Medical Center, AFP Medical Center and Davao Regional Hospital in Tagum, Davao del Norte.
After Legaspi received the shot, Filipina actress Saab Magalona recalled how the doctor saved the life of her son after performing two brain surgeries.
Aside from Legaspi, Food and Drug Administration (FDA) Director General Eric Domingo and health department adviser Dr Edsel Salvaña also received their first shot of the Sinovac vaccine.
The kickoff of the mass vaccination drive in the Philippines on Monday, March 1, comes as health workers express reluctance to receive the Sinovac vaccine.
Domingo earlier said that CoronaVac’s lower efficacy of 50.4% from trials in Brazil led experts not to recommend its use in the priority sector. The Brazil trials involved health workers exposed to COVID-19.
FDA instead recommended its use to people aged 18 to 59 after separate Phase 3 trials in Indonesia and Turkey demonstrated an efficacy of 65.3% to 91.2%. These trials were done mostly in community settings and not among health workers who were exposed to COVID-19.
The recommendation spurred days-long deliberations among experts belonging to the National Immunization Technical Advisory Group, who later approved the use of the vaccine for health workers, reiterating it is safe and effective. – Rappler.com
Add a comment
How does this make you feel?





There are no comments yet. Add your comment to start the conversation.