SUMMARY
This is AI generated summarization, which may have errors. For context, always refer to the full article.
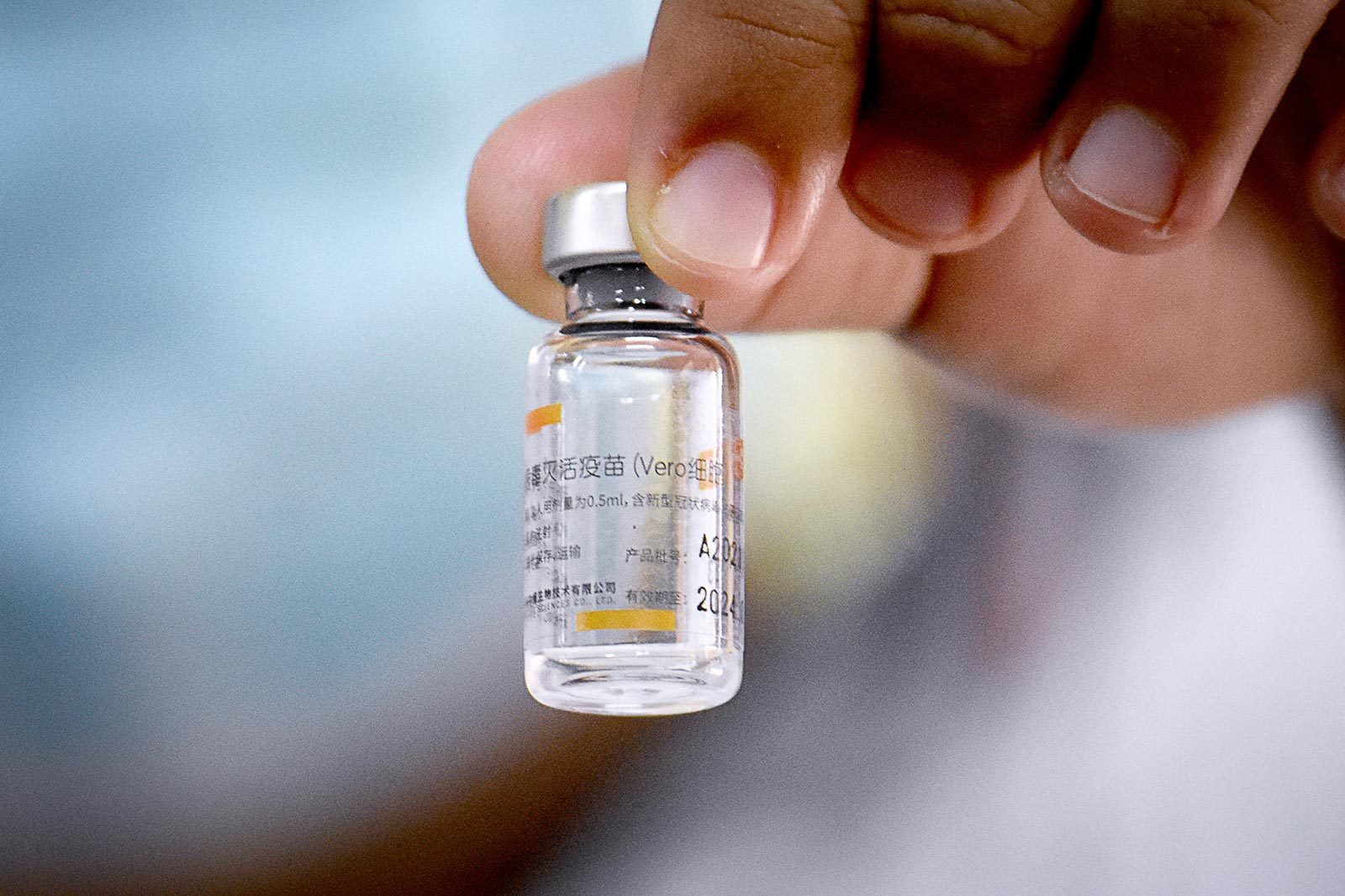

The Philippines’ Food and Drug Administration (FDA) has started looking into whether or not Sinovac’s COVID-19 vaccine can be used for children aged three to 17.
This comes after Sinovac submitted a request to amend its emergency use authorization (EUA) to allow its vaccine’s use among kids.
“Today, just this afternoon, they already applied for an amendment of their EUA to include children, in this case children from three to 17 years old, so this is another potential vaccine that can be used for the pediatric age group,” said FDA Director-General Eric Domingo on Tuesday night, July 6.
He shared this in his briefing on vaccines to President Rodrigo Duterte, during Duterte’s weekly public address about pandemic concerns.
Domingo said the country’s vaccine experts may come up with a final decision on Sinovac’s request within the month of July.
China allowed the use of the Sinovac vaccine among kids in early June, with the company saying the jab successfully triggered an immune response among the age group with only mild adverse reactions reported.
If approved, the Sinovac vaccine would be the second COVID-19 vaccine to be allowed for use among children in the Philippines. In early June, the FDA already allowed the use of the Pfizer-BioNTech vaccine among 12- to 15-year-olds. – Rappler.com
Add a comment
How does this make you feel?


![[Time Trowel] Evolution and the sneakiness of COVID](https://www.rappler.com/tachyon/2024/02/tl-evolution-covid.jpg?resize=257%2C257&crop=455px%2C0px%2C1080px%2C1080px)


There are no comments yet. Add your comment to start the conversation.