SUMMARY
This is AI generated summarization, which may have errors. For context, always refer to the full article.
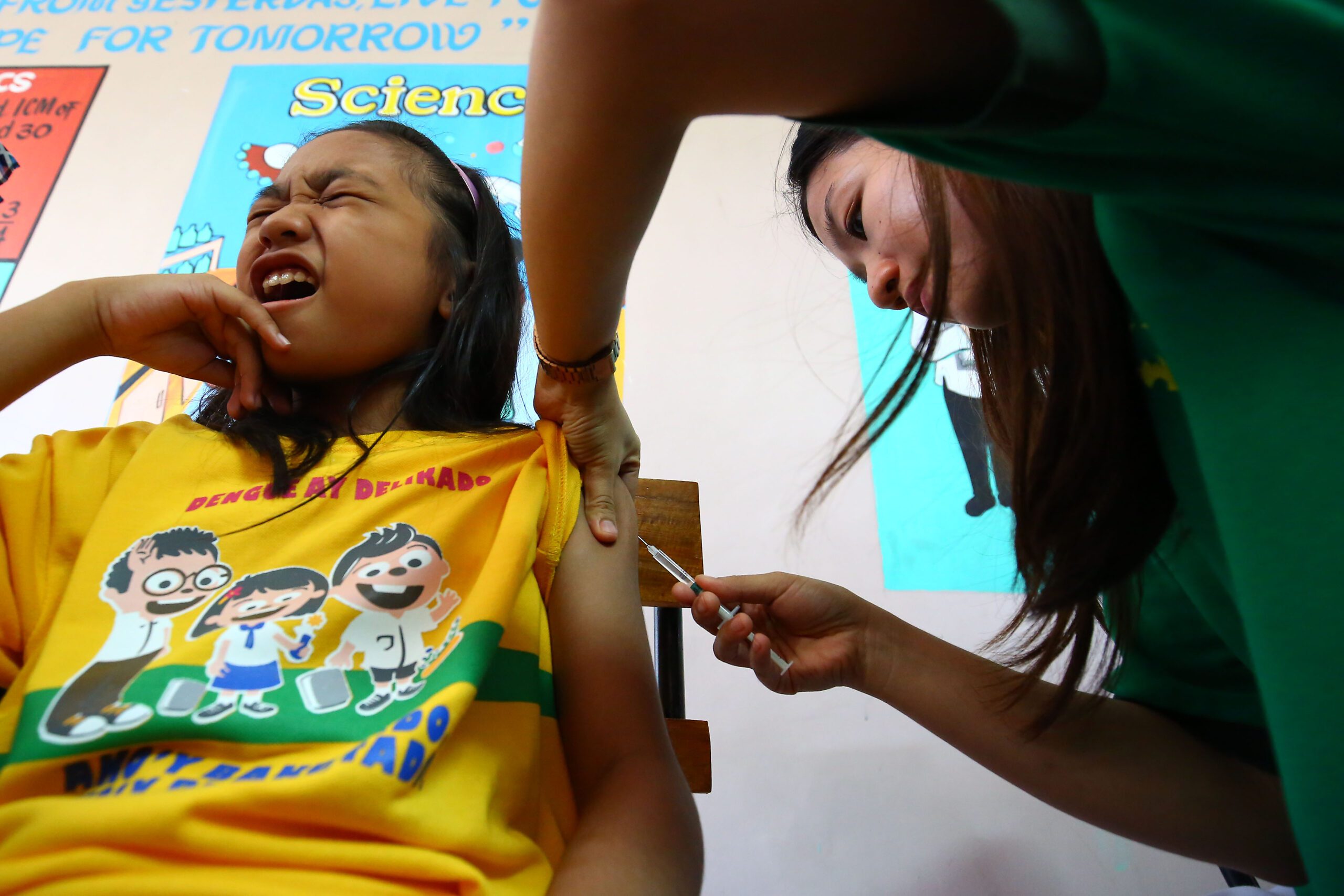
MANILA, Philippines – Less than 4 months after the Philippines approved the sale of the world’s first-ever dengue vaccine, the country’s health department has already begun immunizing over one million Grade 4 students from 3 regions that recorded the highest number of dengue cases in recent years.
Health Secretary Janette Garin led the launch of the school-based dengue immunization program on Monday, April 4, at Parang Elementary School in Marikina City.
“We are the first country to introduce, adopt and implement the first-ever dengue vaccine through public health system and under public school settings,” Garin said during the launch. “With this breakthrough, we can now expand our immunization services to address a disease that is of public health importance.”
While the World Health Organization (WHO) has yet to give its recommendations on the vaccine’s use and public health utility, the Philippines’ Department of Health (DOH) has already vouched for its safety and efficacy.
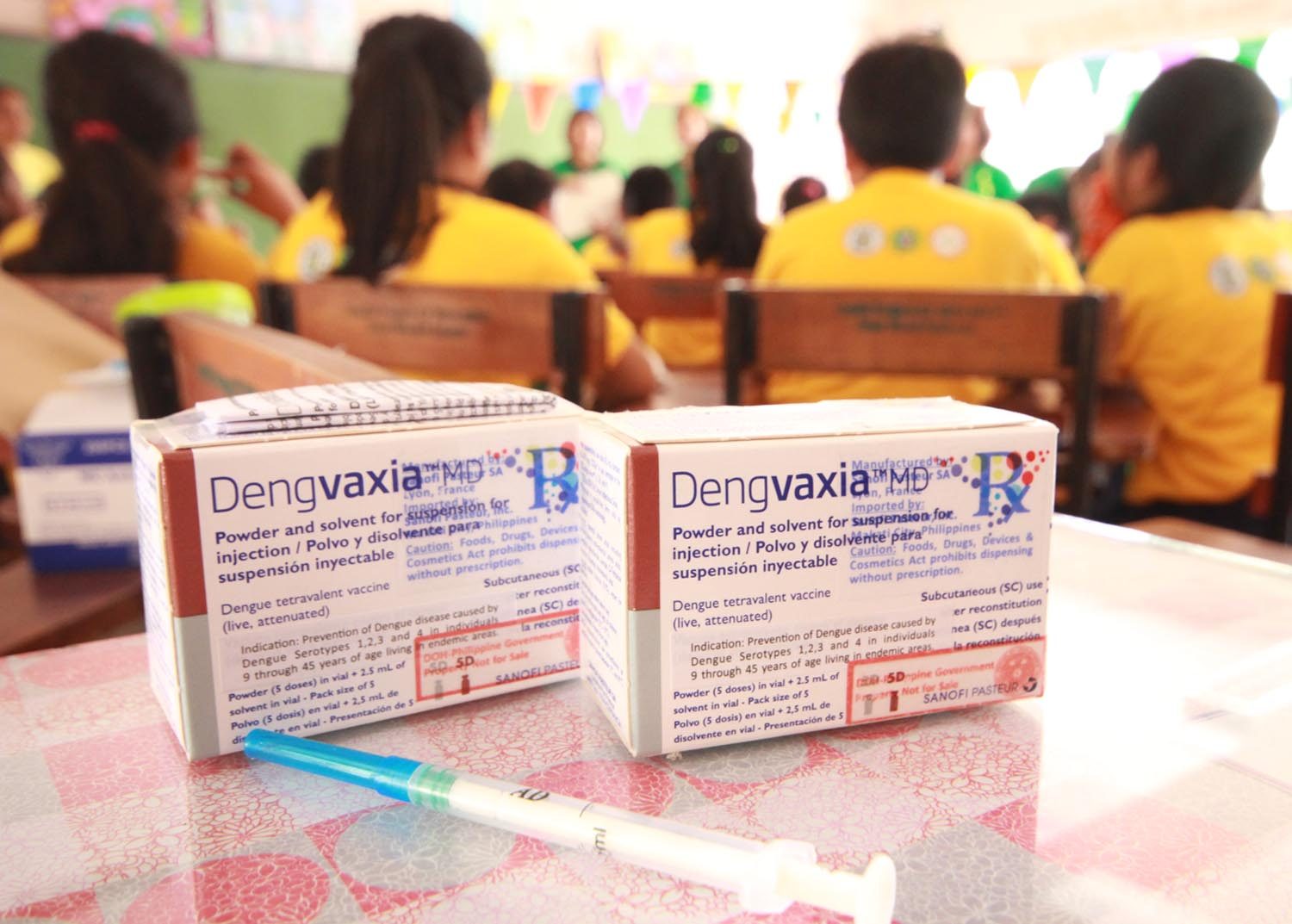
In fact, the department allocated P3.5 billion from its savings to buy the vaccine that will be given to 9-year-old public school students in the National Capital Region, Central Luzon, and Calabarzon.
The first dose of the dengue vaccine will be given from April to June 2016. The same students will then get the second and third doses at 6-month intervals – on October to December 2016 and April to June 2017, respectively.
Dengue, a disease common in tropical and sub-tropical countries, is transmitted through the bite of an Aedes mosquito. Dengue fever is potentially fatal and mainly affects children.
The Philippines is among countries in the Western Pacific region with the highest incidence of dengue in recent years. As of February 20, a total of 18,790 suspected dengue cases were reported nationwide – 13.2% higher compared to the same period in 2015.
‘Unusual’
But health advocates on Monday raised alarm over what they call a “rushed” implementation of the program. (READ: DOH: Vaccine an ‘additional strategy’ to fight dengue in PH)
Dr Anthony Leachon, independent director of the Philippine Health Insurance Corporation (PhilHealth) and a representative of the Monetary Board, said it is “unusual” for the mass vaccination to immediately follow the vaccine’s regulatory approval.
He also asked why the DOH could not wait two more weeks for the recommendation of the WHO’s Strategic Advisory Group of Experts (SAGE) on Dengvaxia.
Leachon, who has 20 years of experience in pharmaceutical medicine, said the usual practice is for the Philippines to wait first for other countries like the United States and Europe, since these countries have “rigid monitoring.”
Besides, he said, if the department waits one more year, the final results of the study will give definite information on the dengue vaccine. Waiting for 3 more years for WHO prequalification, he said, will increase the vaccine’s affordability.
Leachon also raised concerns on how cost-effective the vaccine is.
“Medyo nabahala ako ng kaunti kasi P3.5 billion po ito…. Alam niyo po ba na ang immunization natin sa 7 na gamot ay nagkakahalaga po ng P3 billion, [for diseases such as] measles, mumps, rubella, pneumonia. Ang performance natin 60%. Gagastos ka ng P3.5 billion sa isang sakit na dengue, samantalang ‘yung expanded [program on] immunization na ginawa since 1970s ay hindi natin perpekto.”
(I’m a little concerned about the P3.5 billion…. Did you know our immunization on 7 medicines amount to P3 billion, for diseases such as measles, mumps, rubella, pneumonia. Our performance is 60%. You will spend P3.5 billion for one disease, that is, dengue, when we have yet to perfect our expanded program on immunization established in the 1970s.)
Chances of severe dengue?
Leachon, president of the Philippine College of Physicians Foundation, joins Dr Antonio Dans, who has been vocal against the department for pushing through with the program even without WHO recommendations and prequalification.
Dans, president of the Philippine Society of General Internal Medicine and professor at the University of the Philippines College of Medicine, earlier expressed his worry over “antibody dependent enhancement” – a phenomenon wherein the vaccine might lower the number of dengue cases initially, but could later increase the severity of the disease.
He said severe illness could have a long-term effect on a child’s physical and mental health.
Dans said he may not be a vaccine expert, but he is a clinical epidemiologist who can analyze results of trials.

He believes the vaccine’s safety is not proven because studies on it are stil ongoing. He also pointed out that DOH established intensified surveillance to monitor adverse side effects, and even asked the students to get their parents’ informed consent.
“Kami kampi sa dengue vaccine. Sana manalo ang dengue vaccine. Sino bang kokontra noon? Pero bakit natin sasabihing mananalo tayo kung nasa 1st half pa lang tayo?” Dans said, likening study on the vaccine to a basketball game.
(We side with the dengue vaccine. We hope the dengue vaccine works. Who will oppose that? But how can we say we have already won when we’re just in the 1st half?)
Based on his own analysis, he said he is “97% sure” the WHO will recommend the need to study the dengue vaccine longer.
If the dengue vaccine does not turn out to be as safe and effective as promised, Leachon and Dans are both worried Filipinos will lose their trust in the government’s entire expanded program on immunization. – Rappler.com
Add a comment
How does this make you feel?
There are no comments yet. Add your comment to start the conversation.