SUMMARY
This is AI generated summarization, which may have errors. For context, always refer to the full article.
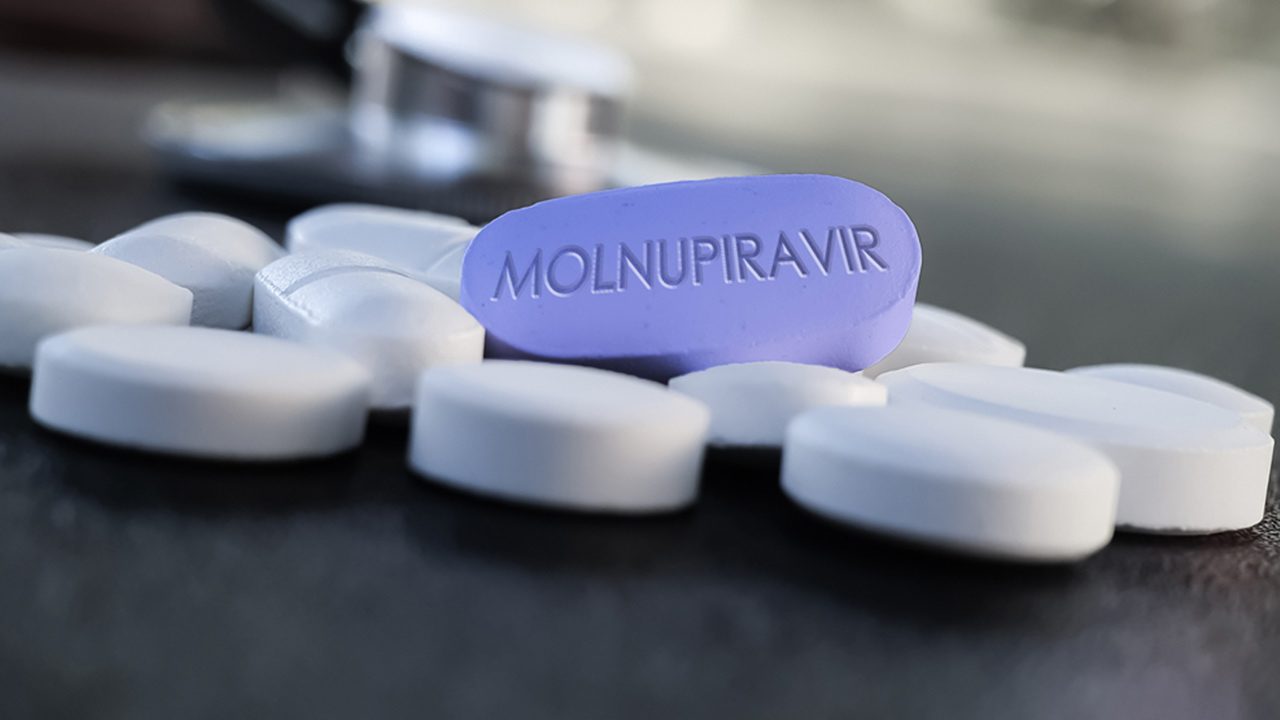
The Lung Center of the Philippines (LCP) and the Quirino Memorial Medical Center (QMMC) are looking for patient referrals who can be included in Phase 3 clinical trials of molnupiravir, an anti-viral drug developed to potentially treat COVID-19 patients with mild to moderate symptoms.
In a press conference on Tuesday, August 17, the two hospitals described the criteria for patients’ inclusion and how the trials work.
“It would be best if we still protect ourselves and prevent getting infected. But if we do get infected, if someone you know…gets infected, we have this promising new drug we can provide to them, which hopefully can improve and hasten their recovery,” said clinical trial investigator Joel Santiaguel of the QMMC.
The hospitals aim to enlist 35 patients in total – 25 for LCP, and 10 for QMMC.
The LCP began its trial on May 17, and has completed the trial for 15 patients. It is looking to recruit the remaining 10. Meanwhile, the QMMC needs the full 10, as it was just recently approved to start its trial.
Molnupiravir is a new drug under investigation as a treatment for currently sick COVID-19 patients with mild to moderate symptoms. It is intended to prevent a mild infection from progressing into a severe case with need for hospitalization.
So far, infectious disease specialists Santiaguel and Virginia delos Reyes of the Lung Center of the Philippines are the only members of the trials’ investigating team, according to the Department of Science and Technology (DOST).
Who can join?
To qualify for the molnupiravir trial, you must satisfy the following criteria:
- At least 18 years old
- Tested positive for COVID-19
- Have had at least one of the following symptoms of COVID-19 (must be mild to moderate) for five days or less before entering the trial
- Cough
- Sore throat
- Nasal congestion
- Runny nose
- Shortness of breath or difficulty breathing
- Muscle or body aches
- Fatigue
- Fever of 38 degrees Celsius
- Chills
- Headache
- Nausea
- Vomiting
- Diarrhea
- Loss of smell or taste
- Have at least one comorbidity
- Not vaccinated against COVID-19
- Not hospitalized
Those with severe symptoms are excluded from the trial.
According to Dr. Loysa Orense, who leads BantAI COVID, qualified patients’ respiratory rate should not be above 30 per minute. Additionally, the heart rate should be less than 125 beats per minute, and the oxygen saturation should be 93 or more.
BantAI COVID is an artificial intelligence-based management system for COVID-19 in Quezon City which helps refer patients for the molnupiravir trials.
How to join
If you think you or someone you know may be qualified for the clinical trial, you can contact the following:
- LCP – Dr. Virginia delos Reyes at 0917-899-9610
- QMMC – Dr. Joel Santiaguel at 0917-841-3314
Delos Reyes from the LCP said that, during the trial, patients will be monitored from their homes. Santiaguel said patients will be monitored until the seventh month from the beginning of the trial.
“Our team makes sure they’re okay. It’s not just the drug, but the care we provide,” said Delos Reyes.
What the results have shown so far
Asked by reporters about the initial results of the LCP’s trial, Delos Reyes said that “we’ll have to wait” for the data of all participating countries to be able to determine the drug’s effects.
However, on August 6, Dr. Randy Castillo from the LCP told ANC that the treatment was a “very promising drug” for COVID-19.
Then, the LCP had enrolled 11 of the targeted 25 patients, who received 800 mg of molnupiravir twice a day for five days. Castillo also disclosed that alongside molnupiravir, the patients still received other drugs to treat their symptoms, like antibiotics and antitussens.
“So far, out of these 11 patients, 80% to 90%, we’ve seen the potential of this drug. These patients were not admitted, just managed at home,” said Castillo.
Worldwide clinical trials for molnupiravir expect a total enrollment of 1,850 participants. Late-stage data for the clinical trials are expected around September to October.
Molnupiravir is a new drug developed by Merck Sharp & Dohme Corporation (MSD). The DOST listed MSD as the implementer and sponsor of the trials in the Philippines.
According to Castillo, molnupiravir was developed specifically for COVID-19.
MSD, who hosted the press conference, is the same company that manufactured ivermectin, an anti-parasitic drug that is also the subject of international clinical trials that cover the Philippines, and has been granted compassionate use permits in at least 6 hospitals as a potential COVID-19 treatment. – Rappler.com

Add a comment
How does this make you feel?


![[Rappler’s Best] US does propaganda? Of course.](https://www.rappler.com/tachyon/2024/06/US-does-propaganda-Of-course-june-17-2024.jpg?resize=257%2C257&crop=236px%2C0px%2C720px%2C720px)







There are no comments yet. Add your comment to start the conversation.