SUMMARY
This is AI generated summarization, which may have errors. For context, always refer to the full article.
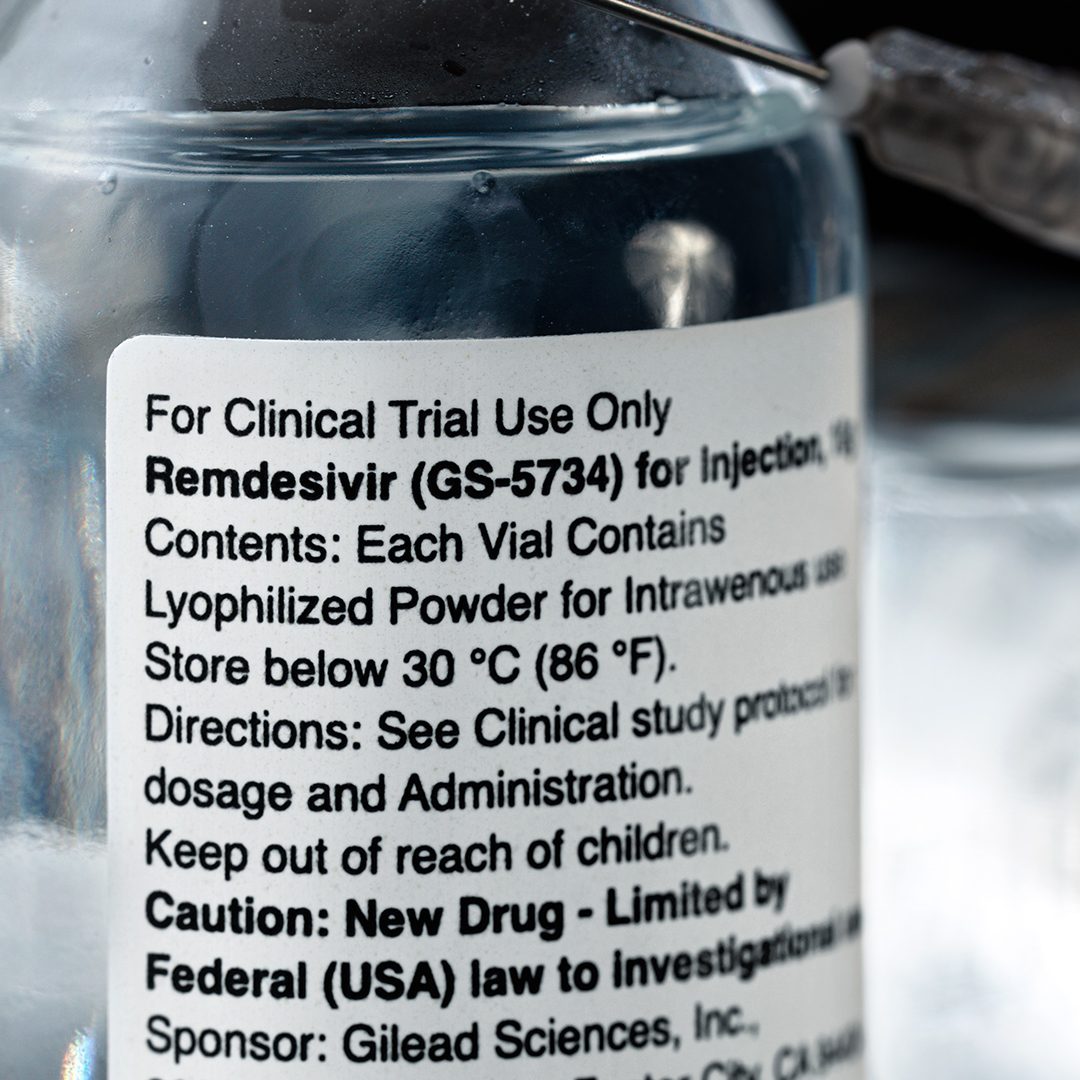
Facing scrutiny from lawmakers, the Department of Health (DOH) defended the use of remdesivir in the Philippines, saying these were covered by compassionate special permits (CSP) granted to hospitals and doctors treating coronavirus patients.
In a press briefing Monday, May 10, Health Undersecretary Maria Rosario Vergeire added the DOH did not procure supplies of the investigational drug, considering it had no emergency use authorization or certificate of product registration (CPR) from the Food and Drug Administration.
“We do not have any procurement of remdesivir, precisely because it has no CPR…. What is being used by hospitals or doctors here is till through the compassionate special permit that these hospitals and doctors applied for,” she said in Filipino.
Vergeire made the statement in response to lawmaker’s claims over the weekend that the DOH was “wasting a ton of money” and could face criminal charges for the purchase of remdesivir supplies in the country.
Anakalusugan representative Michael Defensor had earlier slammed the purchase of the drug as “reckless,” citing the World Health Organization (WHO) recommendation against the drug’s use in COVID-19 patients and its expensive price tag.
Deputy Speaker Lito Atienza echoed this, hitting the DOH’s “double standard” of “promoting” remdesivir, while “stonewalling” other drugs like human-grade ivermectin that was less expensive.
Defensor and Atienza are known advocates of ivermectin, with both claiming during congressional hearings to have used the drug for COVID-19.
Like remdesivir, the FDA granted compassionate special permits to several hospitals to use ivermectin among patients. The FDA likewise recently issued a CPR to a local company to register ivermectin for human anti-parasitic treatment. (As an experimental treatment for COVID-19, a CSP or doctor’s prescription is still needed.)
How remdesivir is used in PH
On Monday, Vergeire said while the WHO did not recommend administering remdesivir in addition to usual care for the treatment of patients hospitalized with COVID-19, other studies, including one conducted in the Philippines by the University of the Philippines National Institutes of Health (UP-NIH) were considered.
Under the UP NIH study, evidence showed the drug could help in slowing down the progression of the disease. The United States, the European Union, and other countries had earlier granted temporary approval for the use of remdesivir after initial research showed it may shorten recovery time in some coronavirus patients.
While the updated treatment guidance was published in the BMJ medical journal last November 2020, the WHO panel had acknowledged that their recommendation does not mean that remdesivir has no benefit for patients.
In the Philippines, Vergeire also said clinical practice guidelines cited remdesivir can be considered as an additional treatment to dexamethasone in COVID-19 patients with oxygen levels below 94%. It did not recommend its use among patients who did not need oxygen supplementation or were already hooked to ventilators.
“We gave funds to hospitals during the time cases were rising so they can have these funds and they can use it to procure whatever investigational drug there is in the market. If they used remdesivir with this money, that is through the CSP. Our hospitals and doctors in the hospitals applied for the CSP to use investigational drugs that do not have a CPR,” Vergeire said in Filipino.
In an interview with ABS-CBN’s TeleRadyo, Health Secretary Francisco Duque III said that the DOH is open to any probe if the lawmakers think there were “irregularities.”
“We’re not hiding anything on that. If there will be an investigation, we welcome it and we will cooperate. There’s no problem with that,” he said in a mix of English and Filipino. – with reports from Bonz Magsambol and Agence France-Presse/Rappler.com
Add a comment
How does this make you feel?


![[Time Trowel] Evolution and the sneakiness of COVID](https://www.rappler.com/tachyon/2024/02/tl-evolution-covid.jpg?resize=257%2C257&crop=455px%2C0px%2C1080px%2C1080px)







There are no comments yet. Add your comment to start the conversation.