SUMMARY
This is AI generated summarization, which may have errors. For context, always refer to the full article.
![[OPINION] On uranium mining and milling](https://www.rappler.com/tachyon/2021/10/uranium-october-14-2021-sq.jpg)
The following is the 16th in a series of excerpts from Kelvin Rodolfo’s ongoing book project “Tilting at the Monster of Morong: Forays Against the Bataan Nuclear Power Plant and Global Nuclear Energy.“
Here, we continue to look at how the fuel for BNPP is produced. It is not mined in the Philippines, because we have none. Perhaps this is just as well, because mining causes serious environmental problems. We do need to know about these difficulties, and acknowledge that if we do go nuclear, people in uranium-producing countries will pay heavy environmental prices.
Traditional mining
Except for special efforts to protect miners from inhaling radioactive dust, traditional uranium mining is not much different from mining for any other metal, as happens all over the Philippines. Deeper deposits are mined underground. If the ore is not too deeply buried, it is cheaper to strip off the overlying soil and rock, and mine at the surface in open pits. In either case, the rock ore is blasted and broken into manageable pieces and brought to a mill at the mine site.
Heavy mill machinery crushes the ore into sand-sized and smaller fragments, and mixes it into a slurry with water. The slurry is then treated with a powerful chemical, most often sulfuric acid. We are familiar with sulfuric acid; it is used in common lead-acid car batteries, and quickly dissolves holes in your clothing if you are careless.
The acid transforms the uranium into crude uranium oxide, or U3O8, called “yellowcake” because of its color. If you are old enough, you may have heard of yellowcake in connection with the US invasion of Iraq in 2003. Dick Cheney, George Bush, and George Blair used forged documentation to claim that Saddam Hussein was buying yellowcake from Niger to make nuclear weapons.
The yellowcake and the weapons turned out to be fictitious, but the hundreds of thousands of people killed by the Iraq war were very real indeed.

About 90 to 95% of the uranium is recovered as yellowcake from the fluid in the slurry. It contains 15 to 25% impurities and still needs more refinement after shipping. And no, it is not radioactive enough to be dangerous to hold like this, but is toxic if you breathe or eat it, so if you ever get the chance to do so, please don’t.
Heap leaching
After having being blasted and quarried out, some ores turn out to contain too little uranium to be milled profitably. A few mines recover uranium fairly cheaply from such ores by “heap leaching.”
First, to prevent leakage, they make pads of clay, sometimes lined with plastic “geotextile.” After the coarsely crushed ore is heaped on top of the pads, sulfuric acid is dripped or sprayed on it. The acid seeps through the ore, dissolves uranium, and oozes down to perforated pipes on top of the pads.
The facilities are cleverly built on slopes so that the acid in the pipes flows down and through a processing plant. Uranium is extracted from the fluid and dried into yellowcake, and the acid is pumped back up to be applied to the heap again.
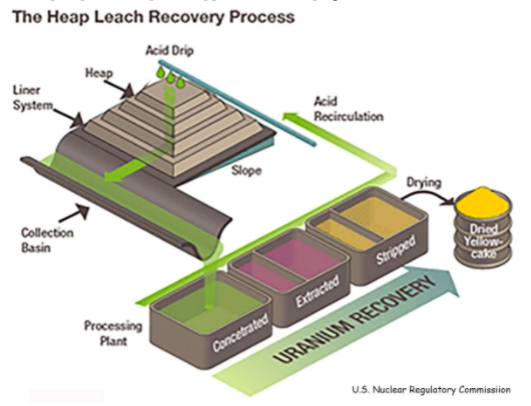
That method is no longer widely used because of serious environmental problems. While active, the heaps emit radioactive radon gas; wind blowing across them disperses dust; the leachate can leak and contaminate streams and groundwater. But even after the leaching is discontinued, the heaps are just another form of mine waste that will pollute the landscape forever.
‘In situ’ leaching: The new way to extract uranium from the ground
Underground and surface mining still provide almost half of the world’s uranium and add to the growing volumes of waste. In the 1970s, however, a new technique, in situ leaching, evolved and rapidly came to dominate the way uranium is produced. One of its main advantages is that it leaves no piles or ponds of waste.
Called in situ because it doesn’t take the ore out of the ground, it pumps sulfuric acid or other chemical fluids into the underground ore deposit through a field of drill holes, and pumps it out containing dissolved uranium through other holes. The uranium is recovered from the fluid as yellowcake on site.
Uranium was first produced by in situ leaching (ISL) in 1975. Its share of uranium production increased dramatically. By 2014 it was 51%, surpassing all other extraction methods. In 2018 it was 55%, largely because the method lends itself well to exploiting the Kazakhstan deposits, which began explosive growth in 2004.
ISL offers many other advantages of over traditional mining. It is cheaper, takes less time to start producing, and uses less energy. The equipment is simpler and easier to maintain. It requires fewer technicians, who are also less exposed to radiation. It disturbs or pollutes the surface environment less, generates much less waste, and can exploit leaner and otherwise inaccessible deposits.

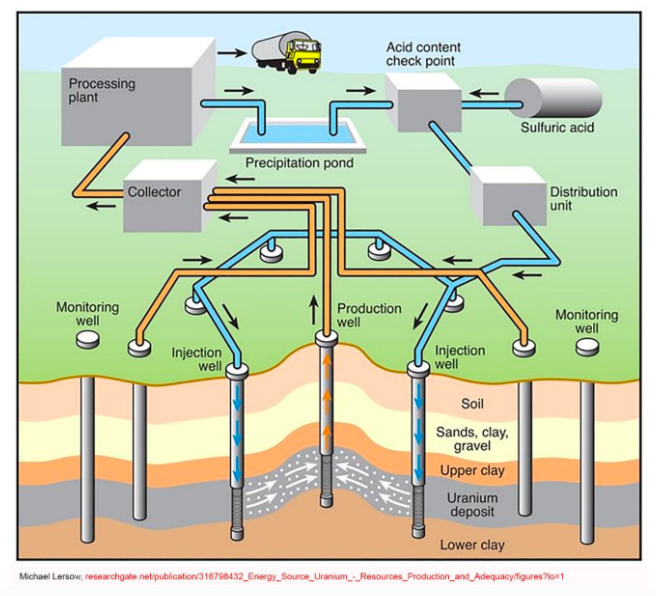
The method does have serious problems. In the United States, the uranium occurs in sandstone layers that also happen to be aquifers that people depend on for their water. Uranium does not dissolve easily in water, but it does in the acid, which disperses it through the sandstone.
In the United States, the Nuclear Regulatory Commission requires ISL operators to get “exemption” permits to degrade the ground-water quality, but also requires them to completely restore the groundwater conditions after extraction ends. That has been impossible to enforce. Most ISL projects have had numerous spills, have contaminated aquifers, and have failed to rehabilitate their wells.
And how do state regulating agencies respond? Not by regulating more strictly; by relaxing their environmental regulations instead. This is the sort of thing that in the Philippines is called “Public Private Partnership.”
Uranium mining waste
Aside from the greenhouse carbon that mining fuel for BNPP makes, huge amounts of mining waste accumulate in piles like these rock “pyramids” left behind when the uranium mine in Ronnenburg, Germany closed.

Elsewhere, milling sludge is stored in ponds, kept wet to avoid radioactive dust from being blown around by wind. These pictured here on the right are at the Olympic Dam mine in Australia. This waste, having been crushed and milled, easily allows rain and melting snow to enter it, weather it, and dissolve radioactive substances, and disperse them into the soils and waters. Like the mining for other, non-radioactive metals, uranium mining also releases heavy metals such as arsenic, and also mill chemicals into the environment.
But the waste from uranium mining poses an additional, even more pressing concern: its radioactivity. A later foray will use the fates of these wastes to examine radioactive decay in detail.
Meanwhile, our next foray will look at the intricate, energy-expensive technology that separates the radioactive U235 from the much less radioactive and overwhelmingly more abundant U238. – Rappler.com
Keep posted on Rappler for the next installment of Rodolfo’s series.
Born in Manila and educated at UP Diliman and the University of Southern California, Dr. Kelvin Rodolfo taught geology and environmental science at the University of Illinois at Chicago since 1966. He specialized in Philippine natural hazards since the 1980s.
Previous pieces from Tilting at the Monster of Morong:
- [OPINION] Tilting at the Monster of Morong
- [OPINION] Mount Natib and her sisters
- [OPINION] Sear, kill, obliterate: On pyroclastic flows and surges
- [OPINION] Beneath the waters of Subic Bay an old pyroclastic-flow deposit, and many faults
- [OPINION] Propaganda about faulting, earthquakes, and the Bataan Nuclear Power Plant
- [OPINION] Discovering the Lubao Fault
- [OPINION] The Lubao Fault at BNPP, and the volcanic threats there
- [OPINION] How Natib volcano and her 2 sisters came to be
- [OPINION] More BNPP threats: A Manila Trench megathrust earthquake and its tsunamis
- [OPINION] Shoddy, shoddy, shoddy: How they built the Bataan Nuclear Power Plant
- [OPINION] Where, oh where, would BNPP’s fuel come from?
- [OPINION] ‘Megatons to Megawatts’: Prices and true costs of nuclear energy
- [OPINION] Uranium enrichment for energy leads to enrichment for weapons
- [OPINION] Introducing the nuclear fuel cycle
Add a comment
How does this make you feel?
There are no comments yet. Add your comment to start the conversation.