SUMMARY
This is AI generated summarization, which may have errors. For context, always refer to the full article.


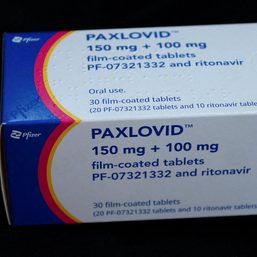
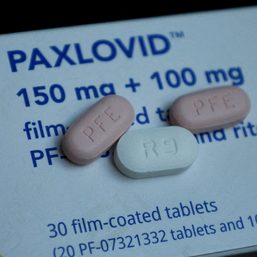
MANILA, Philippines – The Philippines’ Food and Drug Administration (FDA) has granted emergency use authorization (EUA) for Pfizer COVID-19 pill Paxlovid, FDA chief Oscar Gutierrez announced on Friday, March 11.
“Masaya ko pong binabalita sa inyo na naaprubahan ang Paxlovid kahapon. EUA po ‘yun,” Gutierrez said at the Laging Handa press briefing. (We are glad to inform you that we approved Paxlovid yesterday. That’s for EUA.)
“Dalawa na po ang oral anti-viral treatment natin against COVID-19 – Paxlovid and Molnupiravir,” he added. (We already have two oral anti-viral treatment against COVID-19 – Paxlovid and Molnupiravir.)
An EUA would clear the way for products it covered to be used by the public even while these are still in the development phase. COVID-19 vaccines being used now are also under EUA approval.
The granting of EUAs is intended to ensure timely access to medicines and vaccines by cutting down the approval processes that normally take six months, to just about a month.
While an EUA will allow vaccines and medicines to be used by the public, it is not equivalent to a certificate of product registration or authorization to market the product.
Almost 90% effective
The US drug maker in November 2021 said the oral medicine was around 89% effective in preventing hospitalizations or deaths when compared to a placebo, based on interim results in around 1,200 people.
In January, the FDA approved the Department of Health’s compassionate special permit (CSP) for the generic version of Pfizer’s COVID-19 antiviral pill Paxlovid. (READ: FDA approves generic version of Pfizer’s COVID-19 pill)
After reeling from the surge in infections driven by the highly contagious Omicron variant, COVID-19 cases in the Philippines continue to decline, with infections staying below 1,000 for more than a week. – Rappler.com
Add a comment
How does this make you feel?






![[PANOORIN] Naku! Mag-ingat sa unregistered health products online!](https://www.rappler.com/tachyon/2023/05/fact-check-ls-3.jpg?resize=257%2C257&crop=283px%2C0px%2C720px%2C720px)

There are no comments yet. Add your comment to start the conversation.